 Addiction is prevalent all around us, and in particular in the form of illicit substance and alcohol use. Dr. Stacey Daughters’ Biobehavioral Research on Addiction and Emotion (BRANE) lab is interested in understanding the factors that contribute to a substance user’s ability or inability to maintain successful abstinence, and subsequently translating this knowledge into the development and evaluation of novel treatments for substance use.
Addiction is prevalent all around us, and in particular in the form of illicit substance and alcohol use. Dr. Stacey Daughters’ Biobehavioral Research on Addiction and Emotion (BRANE) lab is interested in understanding the factors that contribute to a substance user’s ability or inability to maintain successful abstinence, and subsequently translating this knowledge into the development and evaluation of novel treatments for substance use.
It is known that stress and other forms of negative affect often precede relapse to substance use. Using a computerized behavioral task, Dr. Daughters and her colleagues examine individual differences in the ability to tolerate stress and negative affect, otherwise known as distress tolerance. What their research has collectively demonstrated is that individuals with a substance use disorder who can tolerate the stress and negative affect during this task, or who have high distress tolerance, are significantly more likely to stay in substance use treatment and maintain successful abstinence. 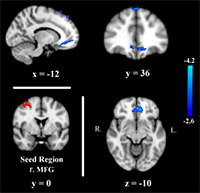 To better understand what might contribute to distress tolerance, they examined the neural process associated with this construct using fMRI. The findings demonstrate that during stress, substance users with higher distress tolerance exhibit stronger connectivity, or co-occurring neural functioning, within areas of their prefrontal cortex, supporting theory that the impairment in the ability of substance users to exert emotion regulation and cognitive control in the face of distress may be a top down neural mechanism.
To better understand what might contribute to distress tolerance, they examined the neural process associated with this construct using fMRI. The findings demonstrate that during stress, substance users with higher distress tolerance exhibit stronger connectivity, or co-occurring neural functioning, within areas of their prefrontal cortex, supporting theory that the impairment in the ability of substance users to exert emotion regulation and cognitive control in the face of distress may be a top down neural mechanism.
 The chronic use of illicit drugs and alcohol also alters the brain’s reward system. As a result, chronic substance users no longer experience the same positive feelings, such as joy and excitement, from everyday non-substance related activities. Dr. Daughters and her colleagues developed a treatment aimed at increasing engagement in non-substance using rewards, called the Life Enhancement Treatment for Substance Use (LETS ACT), and her lab most recently developed a smartphone app for LETS ACT to enable patients to engage in treatment daily. They received a 5 year NIH grant to examine the long term effectiveness of the smartphone enhanced LETS ACT.
The chronic use of illicit drugs and alcohol also alters the brain’s reward system. As a result, chronic substance users no longer experience the same positive feelings, such as joy and excitement, from everyday non-substance related activities. Dr. Daughters and her colleagues developed a treatment aimed at increasing engagement in non-substance using rewards, called the Life Enhancement Treatment for Substance Use (LETS ACT), and her lab most recently developed a smartphone app for LETS ACT to enable patients to engage in treatment daily. They received a 5 year NIH grant to examine the long term effectiveness of the smartphone enhanced LETS ACT.
 In addition to examining the impact of LETS ACT on long term substance use, they will also evaluate how individual differences in neural response to reward predicts treatment success, as well as the effects of the LETS ACT treatment on changing neural responses to reward. Typically, fMRI behavioral tasks examining response to reward use money as a reward. This can be problematic to use with substance users because money is associated with drug craving, making it difficult to know if the results are indicative of reward or drug craving. As such, Dr. Daughters and her lab designed a reward task that utilizes images of people engaging in substance free daily activities that are in line with the LETS ACT treatment approach. Neuroimaging was used to validate this non-monetary reward task in opiate users with elevated depressive symptoms. This task successfully produced biomarkers of reward processing that are typically seen in tasks that use money as a reward in healthy controls and successfully distinguished between opiate users and healthy controls.
In addition to examining the impact of LETS ACT on long term substance use, they will also evaluate how individual differences in neural response to reward predicts treatment success, as well as the effects of the LETS ACT treatment on changing neural responses to reward. Typically, fMRI behavioral tasks examining response to reward use money as a reward. This can be problematic to use with substance users because money is associated with drug craving, making it difficult to know if the results are indicative of reward or drug craving. As such, Dr. Daughters and her lab designed a reward task that utilizes images of people engaging in substance free daily activities that are in line with the LETS ACT treatment approach. Neuroimaging was used to validate this non-monetary reward task in opiate users with elevated depressive symptoms. This task successfully produced biomarkers of reward processing that are typically seen in tasks that use money as a reward in healthy controls and successfully distinguished between opiate users and healthy controls.
 Dr. Daughters is an Associate Professor in the Clinical Psychology program in the Department of Psychology and Neuroscience, faculty in the Human Neuroimaging Group, and UNC Biomedical Research Imaging Center (BRIC). She is also a licensed Clinical Psychologist in the state of North Carolina. Her research expertise includes treatment development for mood and substance use disorders, as well as the identification of neurobiological and behavioral determinants of addiction and associated risk behavior, with a specific focus on distress tolerance and depression.
Dr. Daughters is an Associate Professor in the Clinical Psychology program in the Department of Psychology and Neuroscience, faculty in the Human Neuroimaging Group, and UNC Biomedical Research Imaging Center (BRIC). She is also a licensed Clinical Psychologist in the state of North Carolina. Her research expertise includes treatment development for mood and substance use disorders, as well as the identification of neurobiological and behavioral determinants of addiction and associated risk behavior, with a specific focus on distress tolerance and depression.

