Dr. Sylvia Fitting’s laboratory is interested in sorting the underlying structural and functional consequences of behavior/neurocognition in disease. Her goal is to develop a better understanding of how cellular function is affected by drug abuse ± HIV/HIV-1 protein infection. She focuses on the cellular, functional, and behavioral mechanisms of the neurotoxic consequences of drugs of abuse and HIV-1 Tat interactions on the central nervous system, using state-of-the-art live cell imaging, electrophysiology, and behavioral techniques.
 Human immunodeficiency virus type 1 (HIV-1) infects the brain and, despite combined antiretroviral therapy (cART), many infected individuals suffer from HIV-1-associated neurocognitive disorders (HAND). HAND highly correlates with synaptodendritic damage, including dendritic simplification, axonal disruption, and synaptic loss. The persistence of HAND in the era of cART has raised questions about the causes and treatment of HIV-1-related brain disorders and about the extend to which neuronal dysfunction and injury is reversible. It has been shown that endocannabinoids have protective properties in certain degenerative neurological disorders, including multiple sclerosis, Parkinson, and Alzheimer, by upregulating endogenous cannabinoids anandamide (AEA) and 2-arachidonoyl glycerol (2-AG). Experiments in Dr. Fitting’s laboratory determine the role of the endocannabinoid system in a model of neuro-acquired immunodeficiency syndrome (neuroAIDS) and, by increasing endocannabinoid signaling, determining if it will be protective in models of such neuronal damage.
Human immunodeficiency virus type 1 (HIV-1) infects the brain and, despite combined antiretroviral therapy (cART), many infected individuals suffer from HIV-1-associated neurocognitive disorders (HAND). HAND highly correlates with synaptodendritic damage, including dendritic simplification, axonal disruption, and synaptic loss. The persistence of HAND in the era of cART has raised questions about the causes and treatment of HIV-1-related brain disorders and about the extend to which neuronal dysfunction and injury is reversible. It has been shown that endocannabinoids have protective properties in certain degenerative neurological disorders, including multiple sclerosis, Parkinson, and Alzheimer, by upregulating endogenous cannabinoids anandamide (AEA) and 2-arachidonoyl glycerol (2-AG). Experiments in Dr. Fitting’s laboratory determine the role of the endocannabinoid system in a model of neuro-acquired immunodeficiency syndrome (neuroAIDS) and, by increasing endocannabinoid signaling, determining if it will be protective in models of such neuronal damage.
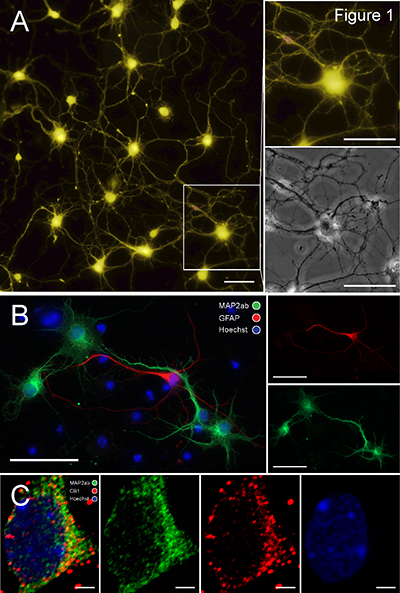
HIV-1 Tat-induced excitotoxicity and neuronal injury/cell death in prefrontal cortex neurons. In Dr. Fitting’s laboratory, researchers use cultured primary dissociated prefrontal cortex neurons as a model to observe Tat effects on neurons (Figure 1). Experiments are performed on cultures at 8–14 days in vitro with neurons forming neuronal networks (Figure 1A, Scale bar, 50 μM). Cultures are characterized by immunohistochemistry (Figure 1B, Scale bar, 50 μM) and contain 92.5% (± 1.64) neurons, assessed by microtubule associated protein 2 immunostaining (MAP2ab, green), and 7.5% (± 1.64) astrocytes, assessed by glial fibrillary acidic protein (GFAP, green). Additionally cannabinoid 1 (CB1) receptors (red) are expressed on neurons (green) in our prefrontal cortex culture (Figure 1C, Scale bar, 2 μM). Previous studies by Dr. Fitting have demonstrated that Tat induces neuron injury, [Ca2+]i production, and behavioral deficits. 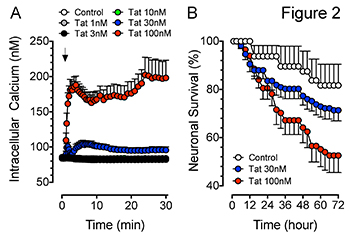 Synaptodendritic injury is a characteristic feature of excitotoxicity and precedes neuronal death. Her in vitro data show a Tat-induced dose response on [Ca2+]i production in our prefrontal cortex neuron culture over a 30 min time period (Figure 2A). Additionally, Dr. Fitting’s results show that Tat induces increased neuronal death in a concentration dependent manner over a 72 hour time period in prefrontal cortex neurons compared to control neurons (Figure 2B). Published data indicate that the Tat-induced excessive Ca2+ influx is accompanied by increases in [Na+]i and mitochondrial instability through a dysregulation of the AMPA/NMDA glutamatergic system. Further, the Tat-induced initial losses in ion homeostasis and increases in [Ca2+]i are attenuated by pyruvate, indicating the involvement of adenosine triphosphate (ATP) depletion due to increases in Na+/K+-dependent ATPase activity.
Synaptodendritic injury is a characteristic feature of excitotoxicity and precedes neuronal death. Her in vitro data show a Tat-induced dose response on [Ca2+]i production in our prefrontal cortex neuron culture over a 30 min time period (Figure 2A). Additionally, Dr. Fitting’s results show that Tat induces increased neuronal death in a concentration dependent manner over a 72 hour time period in prefrontal cortex neurons compared to control neurons (Figure 2B). Published data indicate that the Tat-induced excessive Ca2+ influx is accompanied by increases in [Na+]i and mitochondrial instability through a dysregulation of the AMPA/NMDA glutamatergic system. Further, the Tat-induced initial losses in ion homeostasis and increases in [Ca2+]i are attenuated by pyruvate, indicating the involvement of adenosine triphosphate (ATP) depletion due to increases in Na+/K+-dependent ATPase activity.

Endocannabinoids show neuroprotective effects against HIV-1 Tat-induced excitotoxicity and neuronal injury, mediated via CB1 receptors. No systematic study has been conducted on the neuroprotective effects of endocannabinoid signaling against Tat-induced excitotoxicity and synaptodendritic injury. In Dr. Fitting’s experiments, researchers tested whether applying the direct ligands AEA and 2-AG (3 h pretreatment) can prevent or reverse Tat-induced excitotoxicity/injury/cell death. Cannabinoid receptor antagonists were pretreated 30 min before neuron cultures were exposed to the direct ligands AEA and 2-AG. Data indicate Tat-induced synaptodendritic injury that was significantly reduced by AEA and 2-AG (Figure 3, *p < 0.05 vs. control, #p < 0.05 vs. Tat 100 nM). 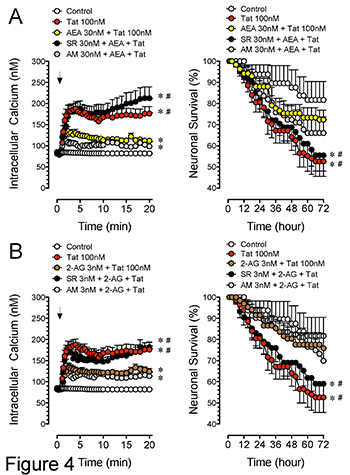 Further, Tat-induced synaptodendritic damage was accompanied by an increase in [Ca2+]i production that was maintained over a 30 min time period and may underlie Tat-induced neuronal death observed over 72 hours (Figure 4). The neuroprotective effects of AEA (decrease in [Ca2+]i and neuronal death) was blocked by CB1 receptor antagonist SR141716A but not the CB2 receptor antagonist AM630 (Figure 4A, *p < 0.05 vs. control, #p < 0.05 vs. AEA + Tat). In contrast, the neuroprotective effects of 2-AG appear to be based on CB1 but not CB2 receptor involvement, as SR141716A blocked the neuroprotective effects of 2-AG, but not the CB2 receptor antagonist AM630 (Figure 4B, *p < 0.05 vs. control, #p < 0.05 vs. 2-AG + Tat).
Further, Tat-induced synaptodendritic damage was accompanied by an increase in [Ca2+]i production that was maintained over a 30 min time period and may underlie Tat-induced neuronal death observed over 72 hours (Figure 4). The neuroprotective effects of AEA (decrease in [Ca2+]i and neuronal death) was blocked by CB1 receptor antagonist SR141716A but not the CB2 receptor antagonist AM630 (Figure 4A, *p < 0.05 vs. control, #p < 0.05 vs. AEA + Tat). In contrast, the neuroprotective effects of 2-AG appear to be based on CB1 but not CB2 receptor involvement, as SR141716A blocked the neuroprotective effects of 2-AG, but not the CB2 receptor antagonist AM630 (Figure 4B, *p < 0.05 vs. control, #p < 0.05 vs. 2-AG + Tat).
Endogenous cannabinoids by itself cannot be used in therapy, as they will be immediately degraded by the main AEA-metabolising enzyme, fatty acid amide hydrolase (FAAH), and of the main 2-AG enzyme, monoacylglycerol lipase (MAGL). Thus, future studies in the Fitting Laboratory will focus on examining the protective effects of endocannabinoid catabolic enzyme inhibitors against Tat neuron injury using the FAAH inhibitor PF3845 and the MAGL inhibitor MJN110. Enzyme inhibitors have high therapeutic potential as they are site specific, meaning they will be produced only at the site of insult and not, as ligands, in other brain areas where it could produce side effects. With enzyme inhibitors the endocannabinoid metabolism is inhibited, thus an accumulation of AEA and 2-AG occurs. Using inhibitors as a tool to study endogenous cannabinoids instead of direct ligands is a quite novel idea and has never been looked at in Tat related neurotoxicity. There is supportive evidence on potential therapeutic applications for inhibitors of endocannabinoid metabolism (FAAH, MAGL). Further, the involvement of the endocannabinoid system in Tat toxicity has not been assessed systematically. Dr. Fitting's goal is to define the mechanisms by which endocannabinoids decrease neuronal injury and synaptic dysfunction in experimental models of HAND and to identify the receptor-mediated pathway that could be targeted therapeutically.

Dr. Fitting is an Associate Professor in the Behavioral Neuroscience Program within the Department of Psychology and Neuroscience at UNC Chapel Hill. Dr. Fitting studies drug abuse and HIV-1 comorbidity by determining the cellular, structural and molecular mechanisms underlying opiod interactions with neuroAIDS. Learn more about her research online.

